TAVI
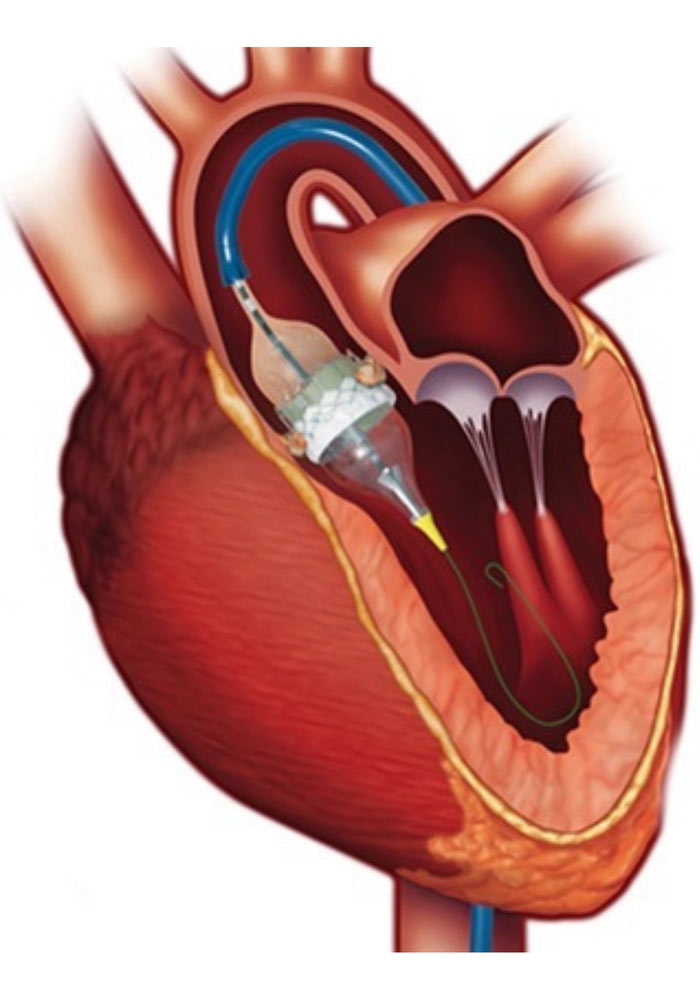
TRANSCATHETER AORTIC VALVE REPLACEMENT(TAVI)
Aortic valve is the valve that connects the heart to the aorta which is the largest artery in the human body that supplies with oxygenated blood to the human body. When we talk about severe aortic valve stenosis we mean that the area of this orifice is restricted from ~ 4 cm2 -which is the normal area- to less than 1 cm2. Symptoms presentation is associated with a poor prognosis (2-5 years) and requires valve replacement. After aortic valve replacement prognosis is restored to that expected for the patient’s status as if he did not suffer from aortic valve stenosis.
Transcatheter aortic valve implantation (transcatheter aortic valve implantation -TAVI- or transcatheter aortic valve replacement -TAVR-) is a revolutionary method that was first performed in 2002 and has since proven its safety and effectiveness. TAVI is used to treat severe aortic valve stenosis. Originally it was utilized as a rescue approach that was applied only to elderly patients who could not undergo the classic “open” surgical replacement of the valve. Surgical aortic valve replacement requires a sternotomy and is performed by a heart surgeon. Extensive clinical trials over nearly two decades have shown that TAVI target populations may be expanded – progressively to younger individuals and at lower risk for surgical replacement.
How is TAVI performed?
The interventional cardiologist inserts a bioprosthetic aortic valve into the body through a peripheral artery, usually the femoral artery – located near the leg -, and then advances it into the “narrowed” (stensosed) native aortic valve. In the final step, a special expansion system is utilized, the bioprosthetic native valve (compressing the structures of the latter). The procedure, depending on each patient’s characteristics and center’s routine, can be performed under only local anesthesia and administration of mild sedation.
Bioprosthetic Devices
The first TAVI interventions urged an explosion of research and development in this field. Today we have at our disposal a range of aortic bioprosthetic “models” produced by a series of specialized biotechnology companies. Aortic bioprostheses consist of leaflets from procine or bovine pericardium and frames constructed by special metal alloys. Depending on the mechanism of their expansion, they are classified as self-expanding or balloon-expanding. They also vary in terms of availability in dimensions, their system of introduction into the human body and their size. This offers flexibility to the interventional cardiologist and the heart team who after extensive preoperative screening decide which bioprosthesis is the most appropriate for each patient.
PreProcedural Evaluation
“Failure to a plan, is a plan for failure.” TAVI procedures have simplified an -otherwise- major surgery for the patient, but not for the medical team. A successful and uncomplicated TAVI requires extensive preoperative evaluation. The patient undergoes a series of examinations such as echocardiography, coronary angiography and CT angiography. These tests enable the heart team to confirm the patient’s anatomical fitness and plan the operation: to decide if percutaneous coronary artery intervention is required – significant lesions in proximal branches are usually treated ad hoc prior to TAVI, to choose the access – ie the route from which the bioprosthesis will be introduced in the body -, to decide on the most appropriate bioprosthesis (type, size) etc.
Post-TAVI
Post interventionally, the patient remains hospitalized for a few days, initially in a unit with the possibility of increased monitoring of vital functions. General progress and increasing experience with the method has significantly reduced postoperative care from ~ 7 days which was in the first years to ~ 2-3 days today in most centers, provided that patient course is uncomplicated.
Risks
TAVI is described as a non-surgical intervention procedure, but this does not mean that it is simple one. The benefits of TAVI far outweigh the risks, but performing TAVI is a major operation with risks. These include access point complications (2 – 30%), stroke (~ 2-3%), need for a permanent pacemaker (2 – 25%), deterioration of renal function and / or acute renal failure (~ 5-6%), peri-procedural acute aortic syndrome and / or death can rarely occur. The wordwide trend is that the frequency of the majority of these complications is gradually decreasing over the years.
